May 9 Update
As the COVID-19 public health emergency (PHE) comes to an end on May 11, I want to remind you of what this means for emergency physicians. Please see this blog for details.
Since I wrote the blog post in February, there have been significant updates to telehealth. See the revised table below for the most up-to-date telehealth policies.
Telehealth Flexibilities
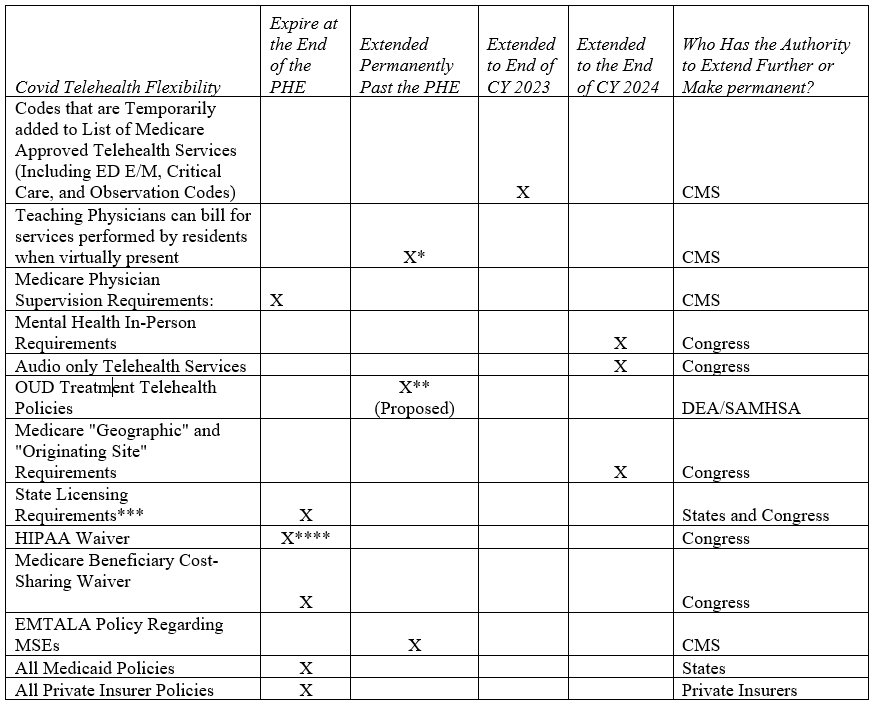 ** Only extended permanently in rural areas.
** Only extended permanently in rural areas.
** Proposed for Opioid Treatment Programs (OTPs) and for buprenorphine prescriptions. However, on May 9, 2023, the DEA announced that as it considers permanent expansions, it will extend the current flexibilities for six months through November 11, 2023.
***Many states have already let this flexibility expire.
**** HHS announced that it was extending its enforcement discretion and will continue not to impose penalties on practitioners that do not use HIPAA-compliant telehealth technology (like skype and facetime) for 90 days post the end of public health emergency (until 11:59 p.m. on August 9, 2023).
___________________________________________________
As I’m sure you’ve heard by now, after three years, the COVID-19 public health emergency (PHE) is coming to an end on May 11. There have been many resources that have already been put together about what this means for physicians and patients—including a great one by the Kaiser Family Foundation. Regs and Eggs has also discussed at length the implications of the PHE ending on telehealth and Medicaid coverage.
However, now that the end date has finally been set, I would like to review the major telehealth and Medicaid policies and then discuss other flexibilities and waivers that you may not realize are expiring as well (let’s face it, three years is a long time, and many of the COVID-19 “temporary” policies have now been baked into our health care system). Before jumping in, it is important to note that some policies, such as the emergency use authorizations for vaccines and therapeutics granted by the Food and Drug Administration (FDA), will not be affected by the end of the PHE.
Telehealth
With respect to telehealth, I previously provided a handy chart that lays out which of the flexibilities will expire at the end of the PHE, and which ones follow a different timeline. Overall, even after the PHE ends on May 11, you will still be able to provide emergency department (ED) evaluation and management (E/M), critical care, and some observation services via telehealth to Medicare beneficiaries through the end of 2023 (but that could be extended!). After those codes are removed from the list, you won’t be able to bill for those services any longer, but you are still allowed to bill office and outpatient E/M codes which are permanently on the list of approved telehealth services.
HOWEVER, although you are allowed to continue billing the ED E/M, critical care, and some observation services through at least December 31, 2023 (and perhaps longer), your ability to provide these emergency telehealth services will be limited going forward. Once May 11 comes along, you won’t be able to provide telehealth services in another state unless you have a license to practice in the state where the patient is located (individual states have different state licensing requirements regarding telehealth, which must be adhered to—and many states have already ended their licensure flexibilities). You won’t be able to use everyday applications such as Facetime and Skype to provide telehealth services. You won’t be able to provide cheaper services to certain Medicare patients in need by deciding not to collect their co-payments, coinsurance, and/or deductibles. You also won’t be able to prescribe buprenorphine for the treatment of opioid use disorder via telehealth unless the Drug Enforcement Administration (DEA) issues a regulation that makes that flexibility permanent.
Thanks to Congress continuing to waive the originating site and geographic Medicare restrictions in the Consolidated Appropriations Act, 2023, you will be able to provide telehealth services to Medicare patients no matter where you or your patient are located (as long as you are in the same state) through the end of calendar year (CY) 2024. Then, starting on January 1, 2025, Medicare telehealth will return to a rural-only benefit, so you won’t be able to provide these services in urban areas. Your Medicare patient must come into an originating site—like a hospital—to receive these services and can no longer receive a telehealth service from their home.
It is important to note that ACEP sent a sign-on letter to the Centers for Medicare & Medicaid Services (CMS) requesting that the agency extend the availability of codes on the Medicare Telehealth List through the end of 2024 to align with the new timeline set by Congress. We were able to get 45 other associations, patient groups, and health care organizations to sign on (thank you!!), and we are hopeful that CMS will listen! However, we still want to be prepared for a scenario where CMS does not act and the ED E/M, critical care, and some observation services are still scheduled to be removed from the Medicare Telehealth Services List at the end of this year. We are therefore planning to send another letter to CMS by February 10 formally requesting that at least some of the emergency medicine codes be permanently added to the Medicare Telehealth Services List.
Medicaid Coverage
As I mentioned previously, there is a significant concern that millions of people will lose their Medicaid and Children’s Health Insurance Program (CHIP) coverage sometime over the next year. To ensure that lower-income Americans received the care they needed during the pandemic, Congress, through the Families First Coronavirus Response Act (FFCRA), provided additional federal support to states that expanded and were able to maintain their Medicaid and CHIP rolls. Nearly all states took advantage of this increased 6.2 percent “Federal Medical Assistance Percentage (FMAP)” in some form—leading to a significant increase in overall Medicaid enrollment.
Once the higher FMAP goes away, states will become responsible for more of the cost of caring for this population. States undoubtedly will need to re-evaluate who will remain eligible for Medicaid as they “unwind” from the PHE and go back to traditional Medicaid and CHIP operations and funding levels. To help states with this “unwinding process” and to ensure that there is a reasonable process in place for reassessing Medicaid eligibility, CMS issued guidance to states that specifically lays out a 12-month time period for making these eligibility re-determinations. CMS is hopeful that some of the people who will be cut from Medicaid will be able to enroll in the Affordable Care Act (ACA) Exchanges.
The enhanced FMAP was initially set to expire at the end of the COVID-19 PHE, but in the Consolidated Appropriations Act, 2023, Congress allowed states to begin processing Medicaid redeterminations this month and disenrolling individuals by April 1 (i.e., before the COVID-19 PHE ends on May 11)— while phasing down the higher FMAP through the end of the year. That means that some of your patients who are currently on Medicaid could lose that coverage sometime in 2023. Data from the Centers for Disease Control and Prevention (CDC) show that individuals covered by Medicaid represented over a third of all ED visits in 2020, although obviously the percentage of patients that you see who are Medicaid enrollees may be different depending on where you practice.
Skilled Nursing Facility 3-Day Rule
Throughout the COVID-19 PHE, a pesky Medicare requirement you probably have heard about (and dislike!) has been waived. In order to for Medicare patients to receive coverage for skilled nursing facility (SNF) extended care services, they must meet the 3-day rule before SNF admission. The 3-day rule requires the patient to have a medically necessary 3-consecutive-day inpatient hospital stay. The 3-consecutive-day count doesn’t include the discharge day or pre-admission time spent in the ED or outpatient observation.
Once the PHE ends on May 11, this waiver will expire, and the 3-day rule will begin to apply again. It is important to note that the 3-day rule will continue to be waived in some of CMS’ demonstration projects through its Innovation Center (CMMI) and in the national accountable care organization (ACO) program, the Medicare Shared Savings Program, even after the PHE ends.
Lastly, I want to mention that the 3-day rule applies to patients in Traditional Medicare. If your patient has Medicare Advantage, the private health plan that enrolls your patient is allowed to require prior authorization before patients can be discharged from a hospital to a SNF. As discussed in a previous blog post, ACEP has heard from our members that this practice has contributed to ED boarding, and we have asked CMS to take action and require Medicare Advantage health plans to end this prior authorization practice.
Free Standing Emergency Department Waiver
Independent free-standing emergency departments (IFEDs) that are not affiliated with hospitals are, under normal circumstances, not eligible to enroll in Medicare and Medicaid. During the PHE, CMS instituted a waiver allowing licensed IFEDs in Colorado, Delaware, Rhode Island, and Texas to temporarily provide care to Medicare and Medicaid patients.
Under the waiver, IFEDs could participate in Medicare and Medicaid in one of three ways:
- Becoming affiliated with a Medicare/Medicaid-certified hospital under the temporary expansion 1135 emergency waiver;
- Participating in Medicaid under the clinic benefit if permitted by the state; or
- Enrolling temporarily as a Medicare/Medicaid-certified hospital to provide hospital services.
Licensed IFEDs choosing to enroll as a hospital during the COVID-19 PHE had to initially meet and continue to meet certain Medicare hospital Conditions of Participation (CoPs). The IFEDs choosing this option have been able to receive hospital facility payments from Medicare based on the inpatient and outpatient care they have provided during the PHE.
Once the PHE ends on May 11, IFEDs who utilized this waiver will lose their hospital billing privileges under Medicare and Medicaid. If a licensed IFED wishes to become a certified hospital after the PHE has ended, it must begin the process of enrollment and initial certification as a certified hospital under the regular processes. An initial survey, either done by the State Agency or Accreditation Organization (like The Joint Commission), will be conducted to determine compliance with all applicable hospital CoPs.
Coverage Rules for COVID-19 Vaccines and Therapeutics
During the PHE, individuals across the country have been able to receive free COVID testing and vaccines. Certain treatments, like antiviral drugs (such as Paxlovid) have also been available without any cost-sharing. This changes once the PHE ends May 11, depending on what type of insurance (or lack of insurance) and individual has. If an individual has Medicare, they will have to start paying for at-home tests, testing-related services, and all COVID-19 treatments. Since March 2020, CMS had been waiving Medicare beneficiaries’ cost-sharing (coinsurance and deductibles) for all services that result in an order for or administration of a COVID-19 test, are related to administering such a test, or to the evaluation of an individual for purposes of determining the need for such a test. This has included ED E/M services, hospital observation services, and office and other outpatient services. CMS had also been completely covering and paying for monoclonal antibody therapies infusions or injections.
Medicaid patients will also likely need to start paying for these services, although specific coverage rules will vary by state. Private insurers, if they haven’t already, will start charging for all these services as well. Further, patients with private insurance (including those enrolled in Medicare Advantage plans) may also start to pay more than they did during the PHE for certain COVID-19 related services delivered by an out-of-network clinician or facility. If a patient doesn’t have health insurance, they will have to pay for COVID-19 testing, vaccines, or treatment. The good news is that patients with any type of insurance should continue to have their vaccinations covered at no cost since vaccinations are considered a preventive service that must be covered without cost-sharing.
With respect to Medicare reimbursement, CMS had been paying clinicians for administering the COVID-19 vaccines at $40 per dose since 2021. This payment rate will continue through the end of this year, and starting in 2024, that rate will drop to $30. CMS will also continue the additional payment of $35.50 for COVID-19 vaccine administration in the home under certain circumstances through the end of the year. Finally, CMS will pay for COVID-19 monoclonal antibodies through this year as well. CMS has been reimbursing $450 for administering a COVID-19 monoclonal antibody in a health care setting, as well $750 for administering a COVID-19 monoclonal antibody therapy in the home. Starting January 1, 2024, CMS will pay physicians administering COVID-19 monoclonal antibody products the same rate as it does for administering other complex biological products.
Liability Protections Under the PREP Act
In order to expand the workforce available and authorized to administer COVID-19 vaccines and other medical “countermeasures” during the PHE, the Administration has used the Public Readiness and Emergency Preparedness Act (PREP Act) to provide immunity to certain medical professionals. Through the PREP Act, immunity has not only been granted to licensed physicians and other health care practitioners, but also to
- Pharmacists and pharmacy interns to administer COVID-19 vaccines (and other immunizations) to children between the ages of 3 and 18, pre-empting any state law that had age limits;
- Healthcare providers licensed in one state to vaccinate against COVID-19 in any state; and
- Physicians, registered nurses, and practical nurses whose licenses expired within the past five years to administer COVID-19 vaccines in any state.
The PREP Act declaration currently expires on October 1, 2024—after which the liability protections will no longer be in place unless a state provides such protections.
Merit-based Incentive Payment System (MIPS)
Since the COVID-19 PHE began in March 2020, CMS has provided some needed relief to Merit-based Incentive Payment System (MIPS) reporting requirements due to the COVID-19 pandemic. MIPS is the major quality reporting program in Medicare for physicians.
CMS provided an automatic exception from MIPS in the 2019, 2020, and 2021 performance periods. With respect to the 2022 performance period, CMS announced on December 29, 2022 that clinicians can continue to apply for an exception from all four MIPS performance categories (Quality, Cost, Improvement Activities, and Promoting Interoperability performance categories) until March 3, 2023. The exception is available for those clinicians whose practice was adversely impacted by COVID-19 and they were unable to meet MIPS requirements because of the response to the disease. Even if you submit an application for an exception, you or your group still have the option of submitting data in two or more performance categories and receiving a performance score. If you do choose to submit enough data to receive a score, your exception is overridden. For more information on submitting a hardship exception for the 2022 performance period, please go to CMS’ website here.
CMS also recently announced that there will be an application-based exception process available for the 2023 performance period. However, given that the PHE is ending on May 11, it may be more difficult to show CMS that you were unable to meet MIPS requirements specifically due to COVID-19. Therefore, the expectation is that most emergency physicians will need to meet MIPS requirements this year.
Other Hospital Policies
During the PHE, hospitals have been able to receive a 20 percent additional bump-up Medicare payment for the treatment of patients diagnosed with COVID-19 through the hospital inpatient prospective payment system. This bump-up payment will end on May 11. ACEP has advocated to CMS that such a bump-up should also apply to physician payments in future public health emergencies.
One popular program that will continue past May 11 is the “Hospital at Home” waiver, which allows hospitals to provide acute care services in a patient’s home. Under the waiver, which has been in place since 2020, CMS does not it enforce its 24-hour on-site nursing requirement for hospitals. To obtain the waiver, hospitals had to complete an application and commit to reporting data on their program to CMS. According to the American Hospital Association (AHA), as of September 2022, 114 health systems and a total of 253 hospitals have been approved to provide hospital at home services to patients. Supporters of this program (like the AHA) have argued that it has helped lead to more effective care, shorter recovery times, and higher patient satisfaction. Congress seems to have agreed with these supporters and decided to extend the waiver through the end of 2024 in the Consolidated Appropriations Act, 2023.
There are also many other blanket waivers and flexibilities that CMS has granted during the PHE, but hopefully I’ve covered the main ones. You will likely have many questions over the next few weeks and months as these flexibilities phase out. We hope that the Administration will put out comprehensive guidance that will help spell out exactly what policies will change and when those changes will occur. In the meantime, if you have any immediate questions, please send them my way!
Until next week, this is Jeffrey saying, enjoy reading regs with your eggs!






