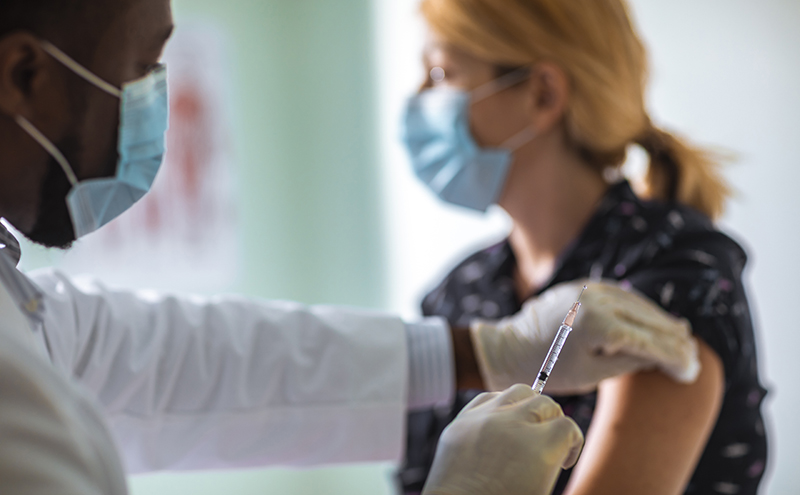
ACEP recommends that all emergency physicians and members of the emergency department staff get the COVID-19 vaccine when it is available to them. By getting vaccinated, you will be leading by example as to the importance and efficacy of the vaccine, as well as reassuring your patients that you are protecting them.
The vaccine details
The vaccine is safe and has been through the complete cycle of trials and scientific rigor that is afforded any other vaccine.
Neither the Pfizer nor the Moderna vaccine are a live virus, but rather made specifically to match the spike protein of COVID-19. (Other vaccines, from AstraZeneca and Johnson & Johnson, that are currently in Phase 3 trials follow the traditional approach of a live-attenuated vaccine. These vaccines should be considered for FDA approval within a few months and it is expected that they will have a similar level of efficacy).
There are two shots required to complete the immunization – one brand 21 days later, the other 28 days later. The vaccines cannot be interchanged – your follow-up shot must be the same brand as the initial shot.
Immunity is expected about 2-3 weeks after the second shot, although a recent study suggested that there may be some immunity as soon as 10 days after the first shot. However, wearing PPE while working clinically and masks in the public is still recommended, as you still could potentially be an asymptotic carrier, and theoretically transmit the disease to others.
There are possible side effects, including fever, sore or swollen arm, and fatigue.
ACEP recommends that emergency physicians schedule their vaccination when they are not scheduled to work on the following day, and also that the ED team staggers their administration of the vaccine over multiple days.
If an emergency physician has had COVID, it is recommended that they still get the vaccine. However, because they may still have some immunity, it may be appropriate for those individuals to wait to be at the end of the line for health care professionals.
The when and how of distribution to emergency physicians
The vaccine from both manufacturers has been distributed to state health departments, which in turn have sent them to local health departments. Some hospitals may already have the vaccines but cannot administer them until the FDA approves the vaccine. Once the FDA approves the use, hospital dissemination plans will begin.
In all states, Group 1A includes emergency physicians, but states can make their own definitions of who is included in this group, which could potentially exclude physicians who are not working clinically, EMS, and other distinctions from receiving their shot through the hospital. However, there should be plans in place to vaccinate members of the medical staff and for those physicians who provide care to patients out of private offices.
ACEP issued a statement about Emergency Physicians’ Priority Access to the COVID-19 Vaccine.
The National Academy for State Health Policy States Plan for Vaccinating their Populations against COVID-19 has a breakdown of each state’s phased plan.
What is the ED’s role with giving the vaccine to the public?
Every emergency physician should consider discussing vaccination with appropriate patients during their visit. ACEP encourages emergency physicians to promote vaccination with their staff members. The emergency physician voice can be a powerful tool to overcome misconceptions and unfounded concerns.
it is still unclear what the emergency department’s role will be in distributing the vaccine to the public. It could be one or a variety of these scenarios:
- Emergency department staff will provide vaccination to all patients they see who are high-risk.
- Emergency department or hospital staff will provide vaccination to family members of high-risk patients, who accompany the patient.
- A public vaccination area may be set up adjacent to the emergency department and ED staff would direct people to that nearby area. Such an area would ideally be staff by non-ED staff.
ACEP is developing documentation on these different scenarios, which will likely be determined at the state level and vary nationwide. Additionally, ACEP will soon have a smart phrase you can copy into your EHR systems to include on patient discharge papers.
If the first shot is given to a patient in the emergency department, there are questions as to how that person will get the second shot to complete the vaccination. Ideally, the vaccine should be administrated by an office-based physician or a pharmacy that has follow-up systems already in place to alert the patient when it is time for the second shot. There will be state systems to follow such patients, with reminders.
To monitor for potential side effects, and to ensure patients get the second shot, individuals who are vaccinated, including health care professionals, will be asked to volunteer for a cell phone-based system V-Safe. After each shot, this system will text the individual every day for 7 days, then weekly for 3 months, then at 6 months and 12 months. The text will simply ask if they were having symptoms or missing work and remind them about their second shot. This is free, but individuals will need to pay for the text if they don’t have a global data plan.
Emergency physicians should expect to see patients with illnesses, health problems and/or symptoms following immunization. Like any other vaccine, reports of these instances will be needed to be submitted to the CDC’s Vaccine Adverse Event Reporting System (VAERS) database. VAERS has a form to fill out that requires patient identification information, takes about 10-15 minutes to complete. The information is HIPPA-compliant, and it is stored in a secure server.
Unfortunately, ACEP expects there may be some members of the public who want to jump the line and could arrive at your emergency department demanding the immunization. ACEP is developing some talking points to help emergency physicians be prepared should this happen.